KEYWORDS
Rhamnogalacturonan-I
cRG-I
Precision prebiotic
Microbiome
Immune
Gut health
Abstract
As a precision prebiotic, cRG-I has the right structural specificity to deliver predictable and consistent effects via the gut ecosystem, stimulating specific health promoting bacteria and increasing the production of beneficial metabolites. Clinical evidence shows that cRG-I can deliver immune and respiratory health benefits. Data also suggests that cRG-I could control inflammation via immunoregulatory mechanisms. Preclinical and clinical studies show how cRG-I increases the abundance of bifidobacteria and other beneficial gut bacteria as well as the production of active metabolites thereby supporting a stable, balanced gut microbiota and healthy gut environment. Emerging evidence supports gut comfort benefits and better barrier function for protection from the outside world and resilience to gut infections. Furthermore, the scientific literature suggests that production of microbial metabolites involved in gut-brain signaling could benefit mood and wellbeing.
Introduction
A nutritious diet not only provides a balanced supply of (micro)nutrients fundamental for good health, but it also influences the composition and function of the microbial communities that colonize the gastrointestinal (GI) tract. While balanced host-microbe interactions modulate biological processes that are essential for health, disruption of this symbiotic interrelationship between the host with its microbiota (dysbiosis) can contribute to the development of various chronic inflammatory diseases like inflammatory bowel disease (IBD), allergies and type-2 diabetes, as is reflected in Western countries where fiber intake is low (1,2). Strategies to modulate the gut microbiota and subsequent host-microbe interactions include the fortification of foods and beverages as well as dietary supplementation with probiotics, synbiotics, postbiotics and/or prebiotics (Table 1).
Table 1. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus definitions (3).

Dietary fibers and prebiotic ingredients
Dietary components like fiber can increase the abundance of beneficial microorganisms and the production of active metabolites like short chain fatty acids (SCFA), B vitamins and precursors for neurotransmitters. Various parameters are used to classify dietary fibers, and these include the primary food source, fermentability, water solubility, viscosity, degree of polymerization (DP) and chemical structure. Common dietary fiber sources include fruit and vegetables, legumes, nuts, seeds, and cereals. Whereas insoluble fibers like (hemi-)cellulose are not, or only slowly, fermented by the bacteria in the human colon and mainly contribute to fecal bulking, soluble fibers are fermented - giving rise to metabolites like SCFAs - without contributing to fecal bulking. Some soluble fibers are viscous and can form gel structures in the gut that can influence post-prandial metabolism by delaying the uptake of dietary sugars and fats. Dietary fibers are mainly carbohydrate polymers and oligomers made up of one or more types of sugar units in 5- or 6-member rings (e.g., fructose, glucose, galactose, arabinose, rhamnose, etc.), bound together through glycosidic linkages that occur at different positions. Additionally, polymers of carbohydrate fibers can be arranged together resulting in structurally complex networks with distinct three-dimensional arrangements (5).
A prebiotic is typically a soluble, non-digestible polysaccharide/oligosaccharide that is not hydrolyzed by host digestive enzymes but fermented by the gut microbiota (3,4). Some of the mechanisms that have been demonstrated to contribute to health benefits (seem to) rely on direct effects of the stimulated microorganisms e.g., bifidobacteria (6,7,8), production of active metabolites like SCFA (9) and precursors for neurotransmitters (10), physiological changes like pH lowering, and protection of the gut epithelial and mucus layer (6,8). Common prebiotics include inulin which is a relatively simple carbohydrate polymer of fructose units with a DP varying from 2 to 60 and linked by β-(2, 1) glycosidic bonds, and smaller oligosaccharides like fructo-oligosaccharide (FOS) with a DP of 2-9 for short chains and 10-60 for long chains, and galactooligosaccharide (GOS) with a DP of 3-8 with β-(1, 4) glycosidic bonds. Next to these structurally simple fibers, more complex non-digestible structures such as pectic polysaccharides, like rhamnogalacturonan-I from carrot (cRG-I), are increasingly described as prebiotics (11).
Hierarchical specificity of dietary fiber
Dietary interventions with fibers aimed to shape the composition and function of the gut microbiota have often presented inconsistent responses. This variability is most likely due to the differences in microbial communities that individuals naturally have. However, it was recently proposed that the structural complexity of dietary fibers could also play a role, and that dietary fibers can be classed in a hierarchical way – based on their specificity. Low specificity fibers are structurally simple and can be easily utilized by many gut bacteria in most individuals, whereas very high specificity fibers are structurally complex (or unique in the diet) but are only utilized by a narrow group of specialized gut bacteria that are not present in all individuals (5). According to this classification, FOS, GOS and inulin which are simple oligomers/polymers of only one or two sugar molecules are classed as low specificity fibers, while xanthan and arabinoxylan as well as insoluble β-1,3-glucan, with unique structures or that are uncommon in the diet are classified as (very) high specificity fibers.
What makes cRG-I a precision prebiotic?
Carrot derived RG-I has a complex molecular structure: it comprises multiple different sugar molecules (galacturonic acid, rhamnose, galactose and arabinose), it has a backbone of repeating disaccharide units (galacturonic acid and rhamnose) and the rhamnose units in the RG-I backbone can be substituted with sidechains like galactans, arabinans or arabino-galactans together comprising more than 15 different glycosidic linkages (12). Studies show how the structural complexity of cRG-I enables the stimulation of selected groups of collaborating (cross feeding) microorganisms present in the gut of most people while well-known prebiotics with relatively simple molecular structures and low specificity, like inulin, can be fermented by a wide range of microorganisms in the gut (13,14). These results illustrate how the structural complexity of dietary fibers determines which bacterial communities can ferment them and benefit from a dietary intervention, thereby supporting the notion that cRG-I is a precision prebiotic. Precise targeting of specific beneficial bacteria common in the core human microbiota could help modulate the microbiota composition and function irrespective of baseline microbiota composition differences, thus potentially leading to consistent health effects in humans (14).
Health benefits via the gut
As cRG-I, which is not digested in the upper GI tract, passes through the small intestine it is sensed by pattern recognition receptors on immune cells that sample the gut content - this first mode of action results in immune training and increased innate immune cell responsiveness. In a randomized double-blind, placebo-controlled study in healthy volunteers, cRG-I was demonstrated to accelerate, and augment innate immune and anti-viral interferon responses to a controlled challenge with a rhinovirus after 8 weeks supplementation (15). Daily intake of cRG-I also enhanced natural killer cell responses ex vivo (16). The immunomodulatory effects of cRG-I resulted in up to 33% reduced severity and up to 3 days shorter duration of symptoms as well as a reduction in the negative effects of a common cold on quality of life (Figure 1) (16).
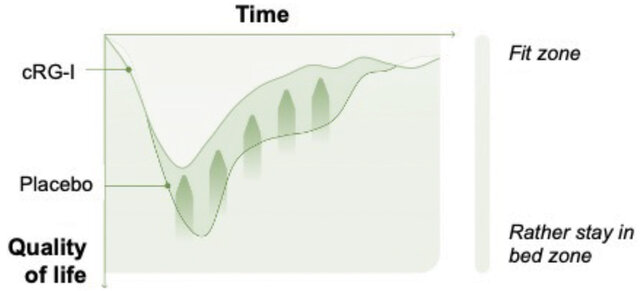
Figure 1. The negative impact of a common cold on quality of life was reduced in subjects taking cRG-I as a daily dietary supplement. (Adapted from Mckay et al 2022, (17)).
The second mode of action involves the modulation of the gut microbiota, i.e., via the selective stimulation of health beneficial bacteria and the increased production of beneficial metabolites by cRG-I.
In the same randomized, double-blind, placebo-controlled dietary intervention in healthy volunteers, microbiota composition analysis was performed on the fecal samples collected at baseline and after 8 weeks supplementation with cRG-I. A dose-dependent increase was observed in the abundance of health beneficial gut commensal bacteria such as Bifidobacterium species (especially Bifidobacterium longum and Bifidobacterium adolescentis) which corroborates in vitro fecal fermentation data (see section below). Significantly more participants in the cRG-I groups had increased levels of Bifidobacterium compared to the placebo group. An increase in the relative abundance of several SCFA producing Firmicutes was also observed (18).
Consistent and predictable modulation of beneficial gut microbes and their metabolites
A recent study using a validated high-throughput ex vivo gut digestion and fermentation simulator (SIFR® technology) compared the prebiotic effects of three dietary fibers of various specificity (i.e., structural complexity) on the intestinal microbiota of healthy adults (14). Fecal samples from 24 subjects were incubated for 48 hours with the high specificity cRG-I fiber (0.3 g and 1.5 g), 1.5 g of inulin (a low specificity fiber) or 1.5 g of xanthan (a very high specificity fiber). It was shown that cRG-I significantly increased a broad spectrum of operational taxonomic units (OTUs, units used to describe bacterial communities), including OTUs of health-related species such as Bifidobacterium longum, Bifidobacterium adolescentis, Anaerobutyricum hallii and Faecalibacterium prausnitzii. The fecal samples from the 24 healthy donors represented the three main human ‘enterotypes’ and the high specificity fiber, cRG-I, selectively stimulated taxa or species of bacteria that were consistently present among human adults, regardless of initial differences in the microbiota composition (Figure 2).
A significant increase in the production of short chain fatty acids (acetate, propionate and butyrate) that play an important role in host-microbiome “cross talk” and that modulate many human bodily functions, including the immune system was also observed in this model (14).
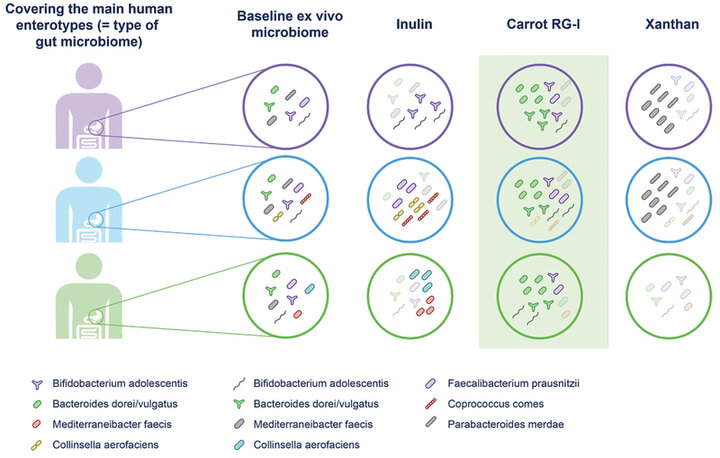
Figure 2. Robust and consistent modulation of beneficial microbes by the high specificity fiber cRG-I across three major enterotypes. (Adapted from van den Abbeele 2023, 14).
Health benefits related to increased abundance of beneficial gut bacteria and microbial metabolites
The health benefits attributed to Bifidobacterium species are numerous and include antipathogenic effects, immune modulation, the prevention of gut disorders, and the production of beneficial metabolites and vitamins (6-9,19,20,21,22,23,24). Concomitant increases in the abundance of anti-inflammatory bacterial species like Anaerobutyricum hallii and Faecalibacterium prausnitzii are aligned with the increased production of SCFA like propionate and butyrate. In addition to supporting immune function and anti-inflammatory effects, SCFA also decrease the pH of the colonic lumen which can increase mineral absorption (25) and inhibit growth of pathogens (26). Health effects related to increased levels of propionate include promotion of satiety, blood cholesterol lowering, reduction of liver lipogenesis and improvement of insulin sensitivity (27). Butyrate on the other hand is the preferred energy source of gut epithelial cells and plays a protective role in gut health, helping to maintain gut barrier function and preventing a leaky gut (21,23).
Less gas production
Fermentation of dietary fibers and prebiotics also results in the formation of gas, like hydrogen (H2), carbon dioxide (CO2), methane (CH4) and hydrogen sulfide (H2S). While this gas production is inevitable, excessive gas production should be avoided as it can lead to bloating and abdominal discomfort or pain. Preclinical studies show that fermentation of cRG-I results in significantly less gas formation than other prebiotic fibers (Figure 3) (14), and clinical studies have shown that regular intake of cRG-I did not lead to gut discomfort due to excessive gas production (15).
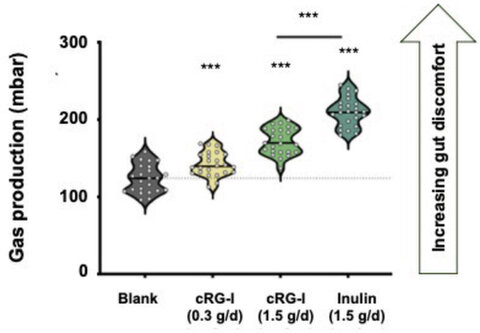
Figure 3. Gas production in the SIFR® model at 48h, n=24. Statistical differences between treatment and blank are indicated with asterisks. (***) padjusted <0.001. (Adapted from van den Abbeele 2023, (14)).
Gut barrier protection
The leaky gut is recognized as an underlying factor for several immune and metabolic diseases as well as a range of psychological disorders (28,29). Increased intestinal epithelial permeability would allow translocation of bacterial cell wall components, metabolites, or even whole bacteria into the systemic circulation, hence contributing to inflammation and injury, not only in the gut but also in remote organs such as the liver and the brain. Therefore, protecting and reinforcing the structure and function of the gut barrier could reduce symptoms of a leaky gut and provide better protection from the outside world as well as more resilience to gut infections. Since fermentation of cRG-I leads to the enrichment of health-associated microbial species and increased production of SCFA, potential effects on gut barrier were investigated using an in vitro model based on the co-culture of intestinal epithelial cells and immune cells exposed to an inflammatory stressor (poke weed mitogen). The supernatant from fecal fermentations in the presence of cRG-I increased epithelial barrier integrity while decreasing markers for inflammation, suggesting that microbial metabolites derived from cRG-I are indeed able to protect a challenged gut barrier (11).
Conclusions
As a precision prebiotic, cRG-I has the right specificity to deliver predictable and consistent effects via the gut ecosystem irrespective of the variability in people’s gut microbiome. Clinical evidence shows that cRG-I can deliver immune and respiratory health benefits - with faster, smarter, and stronger immune responses and increased resilience to airway infections. Data also suggests that cRG-I could control inflammation via immunoregulatory mechanisms leading to faster resolution of (detrimental) inflammatory responses. Preclinical and clinical studies show that cRG-I consistently and in a targeted manner increases the abundance of bifidobacteria and elevates the abundance of other beneficial gut bacteria and the production of their active metabolites thereby supporting a stable, healthy balanced gut microbiota and healthy gut environment (Figure 4). Emerging pre-clinical evidence supports gut health benefits, including gut comfort and barrier function. A more robust barrier could reduce symptoms of a leaky gut and provide better protection from the outside world as well as more resilience to gut infections. Furthermore, the scientific literature suggests that immunoregulatory responses and microbiota modulation could beneficially impact skin health and help manage allergy symptoms, while production of microbial metabolites involved in gut-brain signaling could benefit mood and wellbeing, and optimization of metabolic fluxes could support metabolic health.
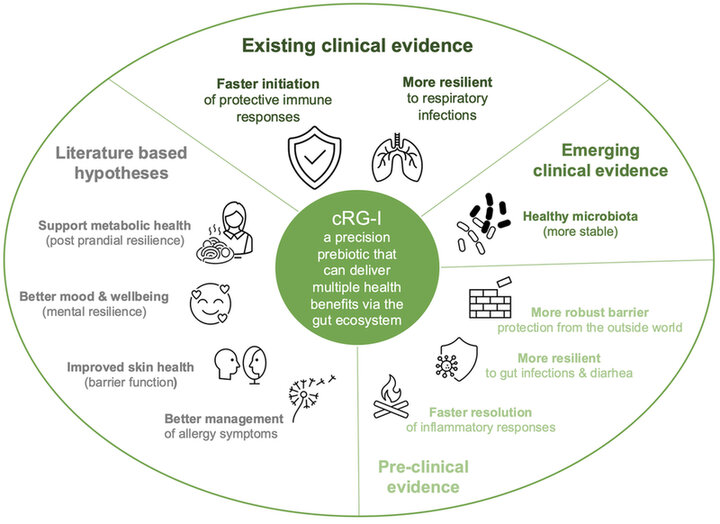
Figure 4. Established and potential health benefits that cRG-I could deliver.



References and notes
- Afzaal, M.; Saeed, F.; Shah, Y.A.; Hussain, M.; Rabail, R.; Socol, C.T.; Hassoun, A.; Pateiro, M.; Lorenzo, J.M.; Rusu, A.V.; et al. Human Gut Microbiota in Health and Disease: Unveiling the Relationship. Front. Microbiol.2022, 13, 999001, doi:10.3389/fmicb.2022.999001. Link Afzaal
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe2018, 23, 705–715, doi:10.1016/j.chom.2018.05.012. Link Makki
- Sanders; M.E. ISAPP Website Available online: https://isappscience.org/a-roundup-of-the-isapp-consensus-definitions-probiotics-prebiotics-synbiotics-postbiotics-and-fermented-foods/ (accessed on 20 February 2024).
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Synbiotics. Nat. Rev. Gastroenterol. Hepatol.2020, 17, 687–701, doi:10.1038/s41575-020-0344-2. Link Swanson
- Cantu-Jungles, T.M.; Bulut, N.; Chambry, E.; Ruthes, A.; Iacomini, M.; Keshavarzian, A.; Johnson, T.A.; Hamaker, B.R. Dietary Fiber Hierarchical Specificity: The Missing Link for Predictable and Strong Shifts in Gut Bacterial Communities. mBio2021, 12, e01028-21, doi:10.1128/mbio.01028-21. Link Cantu Jungles 2021
- Gutierrez, A.; Pucket, B.; Engevik, M.A. Bifidobacterium and the Intestinal Mucus Layer. Microbiome Res. Rep.2023, 2, null-null, doi:10.20517/mrr.2023.37. Link Gutierrez
- Alessandri, G.; Ossiprandi, M.C.; MacSharry, J.; Sinderen, D. van; Ventura, M. Bifidobacterial Dialogue With Its Human Host and Consequent Modulation of the Immune System. Front Immunol2019, 10, 2348, doi:10.3389/fimmu.2019.02348. Link Alessandri
- Schroeder, B.O.; Birchenough, G.M.H.; Ståhlman, M.; Arike, L.; Johansson, M.E.V.; Hansson, G.C.; Bäckhed, F. Bifidobacteria or Fiber Protects against Diet-Induced Microbiota-Mediated Colonic Mucus Deterioration. Cell Host Microbe2018, 23, 27-40.e7, doi:10.1016/j.chom.2017.11.004. Link Schroeder
- Koh, A.; Vadder, F.D.; Kovatcheva-Datchary, P.; Bäckhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell2016, 165, 1332–1345, doi:10.1016/j.cell.2016.05.041. Link Koh
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol Rev2019, 99, 1877–2013, doi:10.1152/physrev.00018.2018. Link Cryan
- Abbeele, P.V. den; Verstrepen, L.; Ghyselinck, J.; Albers, R.; Marzorati, M.; Mercenier, A. A Novel Non-Digestible, Carrot-Derived Polysaccharide (CRG-I) Selectively Modulates the Human Gut Microbiota While Promoting Gut Barrier Integrity: An Integrated In Vitro Approach. Nutrients2020, 12, 1917, doi:10.3390/nu12071917. Link Abbeele 2020
- Kaczmarska, A.; Pieczywek, P.M.; Cybulska, J.; Zdunek, A. Structure and Functionality of Rhamnogalacturonan I in the Cell Wall and in Solution: A Review. Carbohyd Polym2021, 278, 118909, doi:10.1016/j.carbpol.2021.118909. Link Kaczmarska
- Desai, K.; Abbeele, P.V. den; Duysburgh, C.; Albers, R.; Wennekes, T.; Schols, H.A.; Mercenier, A. Structure Dependent Fermentation Kinetics of Dietary Carrot Rhamnogalacturonan-I in an in Vitro Gut Model. Food Hydrocoll.2024, 153, 110036, doi:10.1016/j.foodhyd.2024.110036. Link Desai
- Abbeele, P.V. den; Deyaert, S.; Albers, R.; Baudot, A.; Mercenier, A. Carrot RG-I Reduces Interindividual Differences between 24 Adults through Consistent Effects on Gut Microbiota Composition and Function Ex Vivo. Nutrients2023, 15, 2090, doi:10.3390/nu15092090. Link Abbeele 2023
- Lutter, R.; Teitsma-Jansen, A.; Floris, E.; Lone-Latif, S.; Ravi, A.; Pineros, Y.S.S.; Dekker, T.; Smids, B.; Khurshid, R.; Aparicio-Vergara, M.; et al. The Dietary Intake of Carrot-Derived Rhamnogalacturonan-I Accelerates and Augments the Innate Immune and Anti-Viral Interferon Response to Rhinovirus Infection and Reduces Duration and Severity of Symptoms in Humans in a Randomized Trial. Nutrients2021, 13, 4395, doi:10.3390/nu13124395. Link Lutter
- McKay, S.; Teitsma-Jansen, A.; Floris, E.; Dekker, T.; Smids, B.; Khurshid, R.; Calame, W.; Kardinaal, A.; Lutter, R.; Albers, R. Effects of Dietary Supplementation with Carrot-Derived Rhamnogalacturonan-I (CRG-I) on Accelerated Protective Immune Responses and Quality of Life in Healthy Volunteers Challenged with Rhinovirus in a Randomized Trial. Nutrients2022, 14, 4258, doi:10.3390/nu14204258. Link Mckay 2022
- Mckay, S.; Mercenier, A.; Albers, R. Nutrition and Immunomodulation - Focus on the Dietary Fiber RG-I. Rhamnogalacturonan-I Trains the Immune System and Accelerates Immune Responses. Agro Food Industry hi-tech2022, 33. https://www.teknoscienze.com/Contents/Riviste/Sfogliatore/AF3_2022/6/
- Jian, C.; Sorensen, N.; Lutter, R.; Albers, R.; Vos, W. de; Salonen, A.; Mercenier, A. The Impact of Daily Supplementation with Rhamnogalacturonan-I on the Gut Microbiota in Healthy Adults: A Randomized Controlled Trial. Biomed. Pharmacother.2024, 174, 116561, doi:10.1016/j.biopha.2024.116561. Link Jian 2024
- Derrien, M.; Turroni, F.; Ventura, M.; Sinderen, D. van Insights into Endogenous Bifidobacterium Species in the Human Gut Microbiota during Adulthood. Trends Microbiol.2022, 30, 940–947, doi:10.1016/j.tim.2022.04.004. Link Derrien
- Gonçalves, P.; Araújo, J.R.; Santo, J.P.D. A Cross-Talk Between Microbiota-Derived Short-Chain Fatty Acids and the Host Mucosal Immune System Regulates Intestinal Homeostasis and Inflammatory Bowel Disease. Inflamm. Bowel Dis.2018, 24, 558–572, doi:10.1093/ibd/izx029. Link Goncalves
- Hee, B. van der; Wells, J.M. Microbial Regulation of Host Physiology by Short-Chain Fatty Acids. Trends Microbiol2021, 29, 700–712, doi:10.1016/j.tim.2021.02.001. Link Hee
- Lin, C.; Lin, Y.; Zhang, H.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Intestinal ‘Infant-Type’ Bifidobacteria Mediate Immune System Development in the First 1000 Days of Life. Nutrients2022, 14, 1498, doi:10.3390/nu14071498. Link Lin
- Rivière, A.; Selak, M.; Lantin, D.; Leroy, F.; Vuyst, L.D. Bifidobacteria and Butyrate-Producing Colon Bacteria: Importance and Strategies for Their Stimulation in the Human Gut. Front. Microbiol.2016, 7, 979, doi:10.3389/fmicb.2016.00979. Link Riviere
- Wong, C.B.; Odamaki, T.; Xiao, J. Insights into the Reason of Human-Residential Bifidobacteria (HRB) Being the Natural Inhabitants of the Human Gut and Their Potential Health-Promoting Benefits. FEMS Microbiol. Rev.2020, 44, 369–385, doi:10.1093/femsre/fuaa010. Link Wong
- Whisner, C.M.; Castillo, L.F. Prebiotics, Bone and Mineral Metabolism. Calcif. Tissue Int.2018, 102, 443–479, doi:10.1007/s00223-017-0339-3. Link Whisner
- Yamamura, R.; Inoue, K.Y.; Nishino, K.; Yamasaki, S. Intestinal and Fecal PH in Human Health. Front. Microbiomes2023, 2, 1192316, doi:10.3389/frmbi.2023.1192316. Link Yamamura
- Portincasa, P.; Bonfrate, L.; Vacca, M.; Angelis, M.D.; Farella, I.; Lanza, E.; Khalil, M.; Wang, D.Q.-H.; Sperandio, M.; Ciaula, A.D. Gut Microbiota and Short Chain Fatty Acids: Implications in Glucose Homeostasis. Int. J. Mol. Sci.2022, 23, 1105, doi:10.3390/ijms23031105. Link Portincasa
- Chakaroun, R.M.; Massier, L.; Kovacs, P. Gut Microbiome, Intestinal Permeability, and Tissue Bacteria in Metabolic Disease: Perpetrators or Bystanders? Nutrients2020, 12, 1082, doi:10.3390/nu12041082. Link Chakaroun
- Camilleri, M. Leaky Gut: Mechanisms, Measurement and Clinical Implications in Humans. Gut2019, 68, 1516, doi:10.1136/gutjnl-2019-318427. Link Camilleri


