KEYWORDS
Joint health
Native type II collagen
Functional foods
Marine industry
Sustainable food systems
Regulation
Novel
Joint health has become a public concern due to the high incidence of joint discomfort and mobility restrictions in the oldest populations. Joints are complex structures mainly made up of cartilage, which predominant component is type II collagen. Therefore, research efforts were focused on finding a solution to improve joint mobility and discomfort, positioning native type II collagen as a leading ingredient. Several clinical studies carried out showed its efficacy and safety compared to placebo and even to the standard clinical treatment of chondroitin sulphate and glucosamine chloride. And to top it all off, it can be included in functional foods to make its consumption more accessible to everyone.
Abstract
Introduction
Joint health has become an important public health concern worldwide, largely due to the ageing population. The most common joint condition is osteoarthritis (OA), as it is estimated to affect 32.5 million people only in US (1) and more than 240 million people globally (2).
The use of food supplements to support joint health has gained interest during the last years (3). Among joint health ingredients, collagen has emerged as a focus of particular interest (4), having increased the sales in the US by 26% in 2021 (5). However, the term collagen is being used to define ingredients with totally different structures, compositions, and biological effects (6). This narrative review aims to compile the aspects that define the specificity of native type II collagen to support joint health (Figure 1).

Collagen structure
Collagen superfamily is characterized for containing at least one distinctive triple helical domain. There are 28 types of collagens described differing from each other in their molecular structure, supramolecular organization, and functional characteristics (7). The most predominant ones are from type I to type IV collagens. Type I forms 90% of the total collagen of the body, being relevant in the constitution of skin, bones, teeth, hair and nails. Type II is the main component in the cartilage, creating a cushion that absorbs shocks for joints. Type III is a component of reticular fibres of lungs, liver, dermis, spleen, and vessels while type IV is positioned in the lower membrane layers of the skin and is responsible for filtration (8).
There are different collagen ingredients available in the market containing different forms of collagen: from hydrolysed collagen which is composed by a mixture of amino acids and peptides, to native (undenatured) type II collagen which contains the intact triple-helical structure of the collagen protein (6).
Native (undenatured) type II collagen
Native (undenatured) type II collagen maintains intact the triple helix structure and enhances joint health by means of a specific immune mediated mechanism of action known as oral tolerance (9). Oral tolerance has been defined as the active suppression of specific immune responses to antigens first encountered in the gastrointestinal tract and represents a mechanism to protect from inappropriate immune responses to harmless antigens (10). In summary, antigens are recognized by antigen-presenting cells in the gut-associated lymphoid tissue (GALT) and favour the differentiation of T cells (Tregs) which in turn, modulate the immune response inducing immune tolerance (9). The oral tolerance to type II collagen seems to result in cartilage protection in models of arthritis through the control of the articular inflammation (11, 12). Interestingly, it was demonstrated that when the triple-helix is denatured, this specific mechanism is lost (11). The mechanism of oral tolerance was initially evaluated for autoimmune conditions which is not the case of OA. However, there is evidence suggesting that deregulations of the immune response have an impact on disease pathogenesis (13). In OA, structural changes of joint tissues are driven by mechanical factors and also by inflammation (14). It has been suggested that cartilage fragments could have a role as autoantigens responsible to trigger inflammation (15). In fact, human intervention studies have confirmed the potential of the oral tolerance mechanism to support joint health in different conditions including OA (Figure 2).
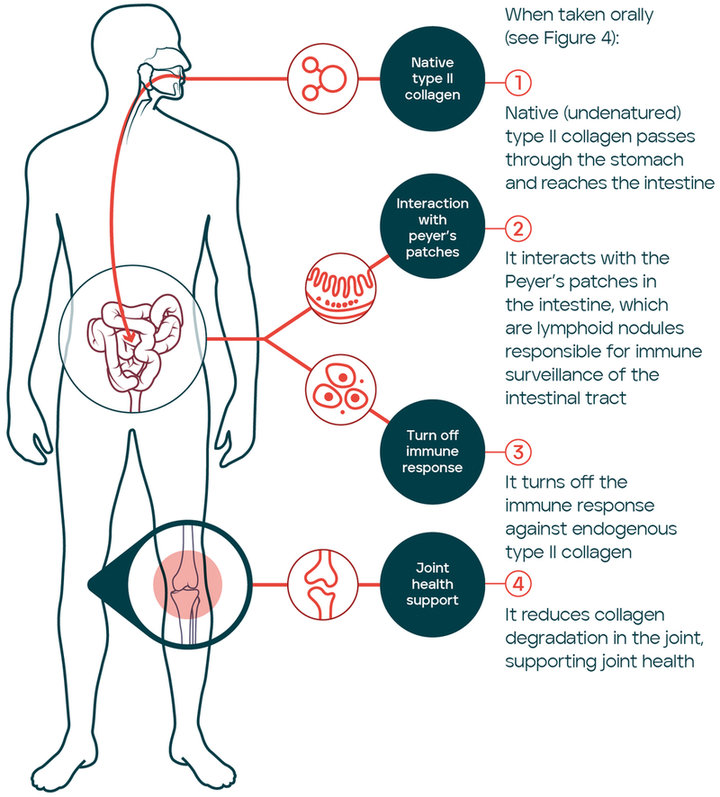
Native (undenatured) type II collagen reaches the Peyer’s patches in the intestine where it turns off the immune response to endogenous type II collagen; reducing collagen degration in the joint and supporting joint health.
Scientific evidence for joint health
Clinical research on supplementation of native (undenatured) type II collagen has mainly focused on its potential therapeutic benefits for joint health. Many randomized, double-blind, placebo-controlled studies showed the improvement on joint mobility and the reduction of joint pain in healthy athletes and runners with knee pain (16-18) and in adults with osteoarthritis (19), compared to placebo. Moreover, a clinical trial compared native type II collagen versus placebo versus the standard clinical treatment of chondroitin sulphate and glucosamine chloride, obtaining remarkable differences compared to placebo and the highest significant improvements in relevant measures of pain (WOMAC score) (20). On the contrary, another study carried out during only 3 months could not see these superior effects (21). Nonetheless, more clinical studies should take place to guarantee the efficacy and safety of native type II collagen and to have more valuable data to ensure it is a valuable treatment to reduce joint discomfort. The most common and accepted parameters to assess the effectiveness are the WOMAC and VAS score, which give a punctuation based on the level of pain of the participants along the clinical study (22).
Interestingly, most of the studies, the tested dose is 40mg/d during a period of 3 to 6 months. This daily dose is low as compared to the dose recommended for hydrolysed collagen (up to 10g/d) (6). For this reason, besides its common use as an ingredient for food supplements, native (undenatured) type II collagen has also been positioned as a potential ingredient for functional foods.
Functional foods
In addition, consumers are looking for alternative formats apart from pills, capsules, and soft gels. The preferred alternatives are gummies, which have grown a 75% in 2021 representing a 21,3% of the non-pill market share (4). Nonetheless, the development of functional foods entails some challenges, such as modifications of the bioactive ingredient due to the conditions encountered during the food manufacturing and extrusion processes, and the development of specific methods of analysis to detect the ingredient in food matrices (23).
Native (undenatured) type II collagen could be especially sensitive to the technological challenges of the food manufacturing processes, because to maintain the biological effects, it is required to preserve the triple helical structure intact. These affecting conditions encompass temperature, humidity, and low pH (24, 25). For instance, during the processing, protein is especially prone to changes in its digestibility and bioavailability (26) and in the extrusion processing, thermal degradation can lead to the denaturation of collagen (27).
In summary, there is a clear opportunity for the development of functional foods containing native type-II collagen, but the nature of the ingredient would require the development of specific production methods to ensure that the ingredient is successfully incorporated into each food matrix preserving its specific biologic activity.
Conclusions
Joint health has become an important public health concern globally. Among joint health ingredients, collagen has emerged as a focus of particular interest. Even though, there are different collagen ingredients with different structures, compositions, and biological effects, native (undenatured) type II collagen is an ingredient characterized by the preservation of triple helix structure. This ingredient has a specific immune-mediated mechanism known as oral tolerance, that inhibits inflammation and tissue catabolism at articular level. Native type II collagen has been shown to be effective in population with joint discomfort at a dose of 40mg/d. The reduced daily dose allows the possibility to include the ingredient in functional foods, besides food supplements.
References and notes
- National institute of aging. November 2022. https://www.nia.nih.gov/health/osteoarthritis
- Hawker GA. Osteoarthritis is a serious disease. Clin Exp Rheumatol. 2019;37 Suppl 120(5):3-6.
- Mordor Intelligence, Bone and joint health ingredients market – growth, trends and forecast (2019-24), [report], 2018.
- Spins Natural Enhanced Channel and SPINS Conventional Multi Outlet (powered by IRI) | 52 Weeks Ending 4-18-2021.
- Nutrition Business Journal 2022, Condition Specific Report
- Martinez-Puig, D., Costa-Larrion, E., Rubio-Rodriguez, N. & Galvez-Martin, P. Collagen Supplementation for Joint Health: The Link between Composition and Scientific Knowledge. Nutrients15 (2023). https://doi.org:10.3390/nu15061332
- Ricard-Blum, S.; Ruggiero, F. The Collagen Superfamily: From the Extracellular Matrix to the Cell Membrane. Pathol Biol (Paris) 2005, 53, 430–442, doi:10.1016/j.patbio.2004.12.024.
- Gelse, K. Collagens—Structure, Function, and Biosynthesis. Advanced Drug Delivery Reviews 2003, 55, 1531–1546, doi:10.1016/j.addr.2003.08.002.
- Park KS, Park MJ, Cho ML, Kwok SK, Ju JH, Ko HJ, et al. Type II collagen oral tolerance; mechanism and role in collagen-induced arthritis and rheumatoid arthritis. Mod Rheumatol. 2009;19(6):581-9.
- Tordesillas L, Berin MC. Mechanisms of Oral Tolerance. Clin Rev Allergy Immunol. 2018;55(2):107-17.
- Nagler-Anderson, C., Bober, L. A., Robinson, M. E., Siskind, G. W. & Thorbecke, G. J. Suppression of type II collagen-induced arthritis by intragastric administration of soluble type II collagen. Proc Natl Acad Sci U S A83, 7443-7446 (1986). https://doi.org:10.1073/pnas.83.19.7443
- Mannelli C, Maresca, M., Micheli, L., Martínez-Puig, D., Ghelardini, C. Low dose chicken native type II collagen is active in a rat model of osteoarthritis. Osteoporosis International. 2015; 26:366.
- Kato T, Xiang Y, Nakamura H, Nishioka K. Neoantigens in osteoarthritic cartilage. Current Opinion in Rheumatology. 2004;16(5):604-8.
- Pelletier JP, Martel-Pelletier J, Abramson SB. Osteoarthritis, an inflammatory disease: potential implication for the selection of new therapeutic targets. Arthritis Rheum. 2001;44(6):1237-47.
- Haseeb A, Haqqi TM. Immunopathogenesis of osteoarthritis. Clinical Immunology. 2013;146(3):185-96.
- Lugo, J.P.; Saiyed, Z.M.; Lau, F.C.; Molina, J.P.L.; Pakdaman, M.N.; Shamie, A.N.; Udani, J.K.; others Undena-tured Type II Collagen (UC-II®) for Joint Support: A Randomized, Double-Blind, Placebo-Controlled Study in Healthy Volunteers. J Int Soc Sports Nutr 2013, 24, 1.
- Schön, C.; Knaub, K.; Alt, W.; Durkee, S.; Saiyed, Z.; Juturu, V. UC-II Undenatured Type II Collagen for Knee Joint Flexibility: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Clinical Study. Journal of Inte-grative and Complementary Medicine 2022, 28, 540–548, doi:10.1089/jicm.2021.0365.
- Shiojima Y, Takahashi M, Takahashi R, Maruyama K, Moriyama H, Bagchi D, Bagchi M, Akanuma M. Efficacy and Safety of Dietary Undenatured Type II Collagen on Joint and Motor Function in Healthy Volunteers: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study. J Am Nutr Assoc. 2023 Mar-Apr;42(3):224-241. doi: 10.1080/07315724.2021.2024466
- Crowley, D.C.; Lau, F.C.; Sharma, P.; Evans, M.; Guthrie, N.; Bagchi, M.; Bagchi, D.; Dey, D.K.; Raychaudhuri, S.P. Safety and Efficacy of Undenatured Type II Collagen in the Treatment of Osteoarthritis of the Knee: A Clinical Trial. International journal of medical sciences 2009, 6, 312.
- Lugo, J.P.; Saiyed, Z.M.; Lane, N.E. Efficacy and Tolerability of an Undenatured Type II Collagen Supplement in Modulating Knee Osteoarthritis Symptoms: A Multicenter Randomized, Double-Blind, Placebo-Controlled Study. Nutrition Journal 2015, 15, doi:10.1186/s12937-016-0130-8.
- Luo C, Su W, Song Y, Srivastava S. Efficacy and safety of native type II collagen in modulating knee osteoarthritis symptoms: a randomised, double-blind, placebo-controlled trial. J Exp Orthop. 2022 Dec 23;9(1):123. doi: 10.1186/s40634-022-00559-8.
- Honvo G, Lengelé L, Charles A, Reginster JY, Bruyère O. Role of Collagen Derivatives in Osteoarthritis and Cartilage Repair: A Systematic Scoping Review With Evidence Mapping. Rheumatol Ther. 2020 Dec;7(4):703-740. doi: 10.1007/s40744-020-00240-5.
- Granato, D. et al. Functional Foods: Product Development, Technological Trends, Efficacy Testing, and Safety. Annu Rev Food Sci Technol11, 93-118 (2020). https://doi.org:10.1146/annurev-food-032519-051708
- Comerford KB, Miller GD, Reinhardt Kapsak W, Brown KA. The Complementary Roles for Plant-Source and Animal-Source Foods in Sustainable Healthy Diets. Nutrients. 2021 Sep 29;13(10):3469. doi: 10.3390/nu13103469.
- Essa MM, Bishir M, Bhat A, Chidambaram SB, Al-Balushi B, Hamdan H, Govindarajan N, Freidland RP, Qoronfleh MW. Functional foods and their impact on health. J Food Sci Technol. 2023 Mar;60(3):820-834. doi: 10.1007/s13197-021-05193-3.
- Hurrell, R.F., Finot, PA. (1983). Food Processing and Storage as a Determinant of Protein and Amino Acid Availability. In: Mauron, J. (eds) Nutritional Adequacy, Nutrient Availability and Needs. Experientia Supplementum, vol 44. Birkhäuser, Basel. https://doi.org/10.1007/978-3-0348-6540-1_9
- Sun, M., Wei, X., Wang, H. et al. Structure Restoration of Thermally Denatured Collagen by Ultrahigh Pressure Treatment. Food Bioprocess Technol 13, 367–378 (2020). https://doi.org/10.1007/s11947-019-02389-6





